Abstract
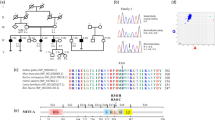
Hereditary non-syndromic profound deafness affects about 1 in 2000 children prior to language acquisition. In 80% of the cases, the mode of transmission is autosomal recessive. The number of genes involved in these recessive forms of isolated deafness (DFNB genes) has been estimated to between 30 and 100. So far, ten DFNB genes have been mapped to human chromosomes, one of which has been isolated2. By linkage analysis of a single family whose members were affected with profound deafness, some of them presenting with vestibular dysfunction, DFNB2 has been mapped to chromosome 11q13 (ref. 3). The gene responsible for a form of Usher syndrome type I, USH1B, has been assigned to the same chromosomal region4. Usher syndrome associates profound congenital deafness and vestibular dysfunction with retinitis pigmentosa. In the homologous murine region are located the shaker-1 mutations responsible for deafness and vestibular dysfunction. It has been demonstrated that the murine shaker-1 and human USH1B phenotypes result from mutations in the gene encoding myosin-VIIA5,6. Based on mapping data as well as on the similarities between the phenotypes of DFNB2-affected patients and shaker-1 mouse mutants, we have proposed that a defective myosin-VIIA may also be responsible for DFNB2 (ref. 1). Sequence analysis of each of the coding exons of the myosin-VIIA gene (MYO7A) was thus undertaken in the DFNB2-affected family. In the last nucleotide of exon 15, a G to A transition was detected, a type of mutation that is known to decrease the efficiency of splicing7â€14. Accordingly, this result shows that different mutations in MYO7A result in either an isolated or a syndromic form of deafness.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Petit, C. Genes responsible for human hereditary deafness: symphony of a thousand. Nature Genet. 14, 385–391 (1996).
Kelsell, D.P. et al. Nature 387, 80–83 (1997).
Guilford, P. et al. A human gene responsible for neurosensory, non-syndromic recessive deafness is a candidate homologue of the mouse sh-1 gene. Hum. Mol. Genet. 3, 989–993 (1994).
Smith, R.J.H. et al. Localization of two genes for Usher syndrome type I to chromosome 11. Genomics 14, 995–1002 (1992).
Gibson, F. et al. A type VII myosin encoded by the mouse deafness gene Shaker-1. Nature 374, 62–64 (1995).
Weil, D. et al. Defective myosin VIIA gene responsible for Usher syndrome type 1B. Nature 374, 60–61 (1995).
Vidaud, M.R. et al. A splice recognition G→C mutation in exon 1 of the human (3-globin gene inhibits pre-mRNA splicing: a mecanism for β+-thalassemia. Proc. Natl. Acad. Sci. USA. 86, 1041–1045 (1989).
Weil, D. et al. Temperature-dependent expression of a collagen splicing defect in the fibroblasts of a patient with Ehlers-Danlos syndrome type VII. J. Biol. Chem. 264, 16804–16809 (1989).
Weil, D., D'Alessio, M., Ramirez, F. & Eyre, D.R. Structural and functional characterization of a splicing mutation in the pro-alpha 2(l) collagen gene of an Ehlers-Danlos type VII patient . J Biol. Chem. 265, 16007–16011 (1990).
Andrews, L.G. & Markert, M.L. Exon skipping in purine nucleoside phosphorylase mRNA processing leading to severe immunodeficiency. J. Biol.Chem. 267, 7834–7838 (1992).
Lind, B., van Solinge, W.W., Schwartz, M. & Thorsen, S. Splice site mutation in the human protein C gene associated with venous thrombosis: demonstration of exon skipping by ectopic transcript analysis. Blood 82, 2423–2432 (1993).
Lemmink, H.H. et al. Mutations in the type IV collagen alpha 3 (COL4A3) gene in autosomal recessive Alport syndrome. Hum. Mol. Genet. 3, 1269–1273 (1994).
Kuivaniemi, H., Tromp, G., Bergfeld, W.F., Kay, M. & Helm, T.N. Ehlers-Danlos syndrome type IV: a single base substitution of the last nucleotide of exon 34 in COL3A1 leads to exon skipping. J. Invest. Dermatol. 105, 352–356 (1995).
Satokata, I., Uchiyama, M. & Tanaka, K. Two novel splicing mutations in the XPA gene in patients with group A xeroderma pigmentosum. Hum. Mol. Genet. 4, 1993–1994 (1995).
Weil, D. et al. Human myosin VIIA responsible for the Usher 1B syndrome: a predicted membrane-associated motor protein expressed in the developing sensory organ. Proc. Natl. Acad Sci. USA 93, 3232–3237 (1996).
Lévy, G. et al. Myosin VIIA gene: heterogeneity of the mutations responsible for Usher syndrome type IB. Hum. Mol. Genet. 6, 111–116 (1997).
Vale, R. Motor proteins, in Guidebook to the Cytoskeletal and Motor Proteins, (eds. KreisT, Vale R) 175–183 (Sambrook and Tooze, Oxford University Press, 1993).
Kyte, J. & Doolittle, R.F. A simple method for displaying the hydropathic character of a protein. J. Mol. Biol. 157, 105–132 (1982).
Rayment, I. et al. Three-dimensional structure of myosin subfragment-1: a molecular motor. Science 261, 50–58 (1993).
Engle, L.J. & Kennett, R.H. Cloning, analysis, and chromosomal localization of myoxin (MYH12), the human homologue to the mouse dilute gene. Genomics 19, 407–416 (1994).
Cheney, R.E. et al. Brain myosin-V is a two-headed unconventional myosin with motor activity. Cell 75, 13–23 (1993).
Vojtek, A.B., Hollenberg, S.M. & Cooper, J.A. Ras interacts directly with the serine/threonine kinase Raf. Cell 74, 205–214 (1993).
Wayne, S. et al. Localization of the Usher syndrome type ID gene (UshID) to chromosome 10. Hum. Mol. Genet. 10, 1689–1692 (1996).
Chaïb, H. et al. Mapping of DFNB12, a gene for a non-syndromal autosomal recessive deafness, to chromosome 10q21-22. Hum. Mol. Genet. 5, 1061–1064 (1996).
Hasson, T., Heintzelman, M.B., Santos-Sacchi, J., Corey, D.P & Mooseker, M.S. Expression in cochlea and retina of myosin Vila, the gene product defective in Usher syndrome type 1 B. Proc. Natl. Acad. Sci. USA 92, 9815–9819 (1995).
EI-Amraoui, A. et al. Human Usher IB/mouse shaker-1: the retinal phenotype discrepancy explained by the presence/absence of myosin VIIA in the photoreceptor cells. Hum. Mol. Genet. 5, 1171–1178 (1996).
Sahly, I., EI-Amraoui, A., Abitbol, M., Petit, C. & Dufier, J.-L Expression of myosin VIIA during mouse embryogenesis. Anat. Embryol. in press (1997).
Tamagawa, Y. et al. A gene for a dominant form of non-syndromic sensorineural deafness (DFNA11) maps within the region containing the DFNB2 recessive deafness gene. Hum. Mol. Genet. 5, 849–852 (1996).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Weil, D., Küssel, P., Blanchard, S. et al. The autosomal recessive isolated deafness, DFNB2, and the Usher 1B syndrome are allelic defects of the myosin-VIIA gene. Nat Genet 16, 191–193 (1997). https://doi.org/10.1038/ng0697-191
-
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ng0697-191
This article is cited by
-
The applications of CRISPR/Cas-mediated genome editing in genetic hearing loss
Cell & Bioscience (2023)
-
Genetic background in late-onset sensorineural hearing loss patients
Journal of Human Genetics (2022)
-
ARNSHL gene identification: past, present and future
Molecular Genetics and Genomics (2022)
-
Identification of autosomal recessive nonsyndromic hearing impairment genes through the study of consanguineous and non-consanguineous families: past, present, and future
Human Genetics (2022)
-
Human deafness-associated variants alter the dynamics of key molecules in hair cell stereocilia F-actin cores
Human Genetics (2022)